What exactly is hepatotoxicity, and why should I be worried?
Hepatotoxicity is damage that occurs to the liver by chemical means. Medications that we use every day can ultimately be damaging our liver!
Unfortunately, hepatotoxicity from commonly known drugs is often a cause of liver injury. It is estimated as many as 1000 drugs can be blamed for some form of liver damage.
The liver’s role in detoxification makes it especially vulnerable to damaging substances. Liver cells, hepatocytes, can be injured by various pathways depending on the type of drug being metabolized. Reactive particles can destruct the cellular components of the hepatocytes, leading to cell death. Other metabolites alter liver enzymes responsible for drug metabolism leading to toxicity. Drug fragments can also bind to liver proteins initiating an autoimmune attack on the liver tissue.
Effects of hepatotoxicity can be seen throughout the body. General symptoms include: nausea, vomiting, upper right stomach pain, loss of appetite, brown urine, and yellowing of the skin and eyes (jaundice.) It is important to note these changes early and notify the health-care provider immediately if they are experienced. Early intervention is the safest way to limit damage to the liver!
Who is at risk for hepatotoxicity? EVERYONE! It may be a patient you are monitoring, a family member you are educating, or yourself. Concerns extend far beyond high dosages of prescription medications. Hepatotoxicity can result from combining multiple drugs, alcohol consumption with medications, genetic predispositions, age-related metabolism rates, and underlying diseases. Over-the-counter products, such as Tylenol, and herbal remedies, that are often considered “safe”, play a HUGE role in liver damage.
The key to lowering your risk is EDUCATION. Knowledge about hepatotoxicity is critical to keeping EVERYONE safe.
Reference:
Kaplowitz, N. (2004). Drug-induced liver injury. Clinical Infectious Diseases: An Official Publication Of The Infectious Diseases Society Of America, 38 Suppl 2S44-8. http://libproxy.uta.edu:2066/.
More information on hepatotoxicity, like specific symptoms to look out for can be found here: http://www.liverdisordersfocus.com/articles/liver-disorders/drug-induced-liver-damage.php
Tuesday, October 27, 2009
The Take Home Message
A valuable resource for information on hepatotoxicity is the FDA!
Investigations into drugs, definative information, and procedures for reporting possible problems associated with a product can be found at http://www.fda.gov/.
Don't forget to ask your health-care provider and pharmacist any questions or concerns!
Over the counter products can cause severe hepatotoxicity! OTC products are not always "safe." Alcohol, liver infections, and drug/food interactions can be dangerous when used with certain OTC products. Accidental overdoses are common!
Acetaminophen is now the number one cause of acute liver failure in the America!
-Many products other than Tylenol contain acetaminophen (OTC and prescribed)
-Read labels. Remember, APAP is the abbreviation for acetaminophen.
-Calculate your dosages. Do not exceed 4000 mg per day (4 grams) normally.
Diet pills have a long history of complications! Consult your health-care provider with their use.
-Take as directed and dedicate yourself to the lifestyle changes that accompany diet therapies.
-Note any changes indicating hepatotoxicity and contact your heath-care provider immediately!
Alcohol changes your normal liver function! Alcohol is a damaging chemical to the liver and can cause greater complications with OTC and prescribed therapies.
Food and drug interactions in the liver are serious!
-Grapefruit interactions can be deadly! Grapefruit leads to hepatotoxicity of many medications. Be aware of drug interactions with grapefruit. You may have to alter your diet!
-Fatty foods may impact the liver's ability to process toxins and fuction normally! A liver riddled with fat may not function efficiently.
Herbal products are not regulated by the FDA and are often linked to liver damage!
Tell your health-care provider when you are using herbal therapy in combination with medications. Be aware of interactions your medications may have with certain supplements, including acetaminophen.
Many prescriptions are well known to induce liver damage, including: Cholesterol lowering medications, Antibiotics, and Antidiabetics.
-Ask your health-care provider about side effects of your medications, specifically in the liver.
-Remember many factors can cause hepatotoxicity of prescription drugs including: age, food interactions, alcohol consumption, and other drug interactions.
Hepatotoxicity will likely continue to be an issue as more medications are developed to battle common health issues. The key to avoiding complications with your liver is to stay in the know about current products as well as your own body changes. Too often, the hepatotoxic dangers lurking in the medicine cabinet are unknown to people. Unfortunately, the realization of these dangers can be a situation of "too little, too late" and serious consequences result.
With this in mind, it raises the question, how can we change this outlook in our community? Is it up to the FDA, the healthcare system, or maybe...YOU?? Who will you tell??
For more information on drug-induced hepatotoxicity, please visit:
http://emedicine.medscape.com/article/169814-overview
http://www.pharmacologyonline.org/PDF/dili.pdf
Saturday, October 24, 2009
Antidiabetic Drugs: The Dark Side
 Approximately 20 million people have diabetes in the U.S. and at least 6 million are unaware of the fact that have diabetes. People with diabetes are predisposed to acute liver injury and are twice as likely to have hepatotoxicity. Antidiabetic drugs can increase liver injury, adding to the complications already impacting the liver.
Approximately 20 million people have diabetes in the U.S. and at least 6 million are unaware of the fact that have diabetes. People with diabetes are predisposed to acute liver injury and are twice as likely to have hepatotoxicity. Antidiabetic drugs can increase liver injury, adding to the complications already impacting the liver. Today, although new medications are available, it is still recommended that healthcare professionals test liver enzymes routinely. As new drugs are developed to treat diabetes it is important to know the side effects, especially those that alter live enzymes. People coping with diabetes should remember there are natural ways to enhance medications and regulate diabetes. Diet and exercise should be done in conjunction with antidiabetic medications.
Can we do more to protect ourselves from these complications? Are antidiabetic drugs that problematic?
For more information on diabetes, please visit:
http://www.diabetes.org/
References:
Vagula, Mary. Sachin S. Devi. Hepatotoxicty of Antidiabetic Drugs. Retrieved October 27, 2009. http://www.uspharmacist.com/c, PhDontent/s/38/c/10086/
Thompson, Cheryl. Antidiabetic Drug Pulled From the Market. Retrieved October 28, 2009. http://www.ashp.org/import/News/HealthSystemPharmacyNews/newsarticle.aspx?id=108
Friday, October 23, 2009
Antiobiotics Fighting Germs: Are They Also Fighting Your Liver?
Hepatotoxicity from these drugs have been known since the 60s when erythromycin was found to have serious effects on the liver in adult patients. Today, a wide variety antibiotics are known to cause liver toxicity including those used to treat staph infections and many penicillins.
How do you know you are at risk while taking an antibiotic? Ask your healthcare provider or pharmacist about potential side effects of your medication. Of course, alcohol use and taking other medications known to alter liver action increases your risk for developing problems. Patients at either end of the age spectrum, either young children or elder adults, are also at an increased risk of problems. Other diseases, such as HIV, AIDS, hepatitis, tuberculosis, and malnutrition, can also increase your risk for developing liver damage with antibiotics.
What is the solution to the problem? EDUCATION for patients is the best way to prevent serious liver damage. Know your risks and take caution when starting an antibiotic treatment. It has been suggested that baseline liver function tests before treatment is key to monitoring changes in the liver. This is a drastic measure that should be utilized in specific situations where infection and the medications used to treat it both play major roles in liver injury (e.g. tuberculosis and hepatitis treatments.)
Had you ever heard of liver damage from antibiotics? Can we do more to protect people from liver injury with antibiotic use?
Reference:
Ybarra, J. (2007). Drugs that harm bugs...and the liver?. Drug Formulary Review, 3-4. http://libproxy.uta.edu:2066/
For more infomation on this topic, please visit:
http://www.antibioticliverdamage.com/
http://www.medicinenet.com/script/main/art.asp?articlekey=94614
Thursday, October 22, 2009
The Big Bad Cholesterol Meds
It is extremely important to note any symptoms you are experiencing with these medications and immediately contact your health-care provider. Also, it is critical that you have your liver function consistently monitored on these medications. Early intervention is key to preventing irreversible damage to your liver!
Are cholesterol medications really as bad as they're made out to be?
References:
Sidharth S. Bhardwaj, MD and Naga Chalasani, MD. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2048990/?tool=pubmed. August 2007.
Kinnman, N., & Hultcrantz, R. (2001). Lipid lowering medication and hepatotoxicity. Journal Of Internal Medicine, 250(3), 183-185. http://libproxy.uta.edu:2066
For more information, please visit:
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2048990/?tool=pubmed
http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm048496.htm#StateoftheStatins
Wednesday, October 21, 2009
Herbal Supplements: Benefit or Risk?
 The use of herbal products has become one of the biggest trends in recent years, as more “natural” drug therapy becomes popular. There are over 29,000 products available on the market. One reason for this is the rise in costs of perscription medication . The public believes that herbal supplements are harmless and reliable for treating or preventing many diseases. A major concern about herbal supplements is the risk of side effects. The FDA does not regulate their use, and the research on them is slim to none. The drugs are placed on the market on a trial and error basis, without any review of the benefits or risks. The producers of herbal supplements are not required to post warnings on labels; this has led to severe liver hepatotoxicity and damage to other organ systems.
The use of herbal products has become one of the biggest trends in recent years, as more “natural” drug therapy becomes popular. There are over 29,000 products available on the market. One reason for this is the rise in costs of perscription medication . The public believes that herbal supplements are harmless and reliable for treating or preventing many diseases. A major concern about herbal supplements is the risk of side effects. The FDA does not regulate their use, and the research on them is slim to none. The drugs are placed on the market on a trial and error basis, without any review of the benefits or risks. The producers of herbal supplements are not required to post warnings on labels; this has led to severe liver hepatotoxicity and damage to other organ systems.Health professionals and the makers of herbal supplements are at odds on this subject. Because of the public view about herbal medication and the "all natural ingredients", use of these supplements has doubled over the last ten years. Between 20% to 30% of consumers have been admitted to a hepatic-clinic for liver injury. Ironically enough, herbal medications seem to be used most by people who have liver diseases.
The risk to the liver can be severe. Cases that have been reported liver injury from mild, emergency transplant, and even death. This can be a surprising fact to most of the public, considering that the consensus of herbal supplements is harmless. In a public survey, 1 out of every 6 consumers report taking an herbal supplement in conjunction with their current prescriptions. This can be a dangerous combination!
There are thousands of stories about this herbal supplement side effects. The major concerns seem to be hepatotoxicity, especially associated with herbal products taken before surgery and use of these medications by the elderly. There are so many herbal products that it is almost impossible to list them along with potential side effects here; however, the number one injury most often cited is to the liver. Hepatotoxicity is at greater risk when acetaminophin and herabal supplements are taking together. Some of the most dangerous herbal drugs to the liver are; Black cohosh, Kava, Echinacea.
The lesson of the day: ask your healthcare provider before combining any drugs, perscriptioin or over-the-counter, with herbal medications.
Are you at risk??
References:
Barger, L. Herbs and Pain Relievers Don't Mix: The Dangers of Mixing Herbal Supplements and OTC Pain Relivers. Retrieved October 22, 2009. http.//herbalmedicine.suite101.com
Larson, A. Hepatotoxicity Due to Herbal Medications. Retrieved October 22, 2009. www.uptodate.com/patients
Is fast-food damaging your liver??

An alarming finding is that these habits are starting to impact our youth. Health-care providers are beginning to see teenagers and even some children with hints of cirrhosis!
Is fast food completely to blame? Excercise is an important part of balancing your caloric intake. This can help minimize the effects of unhealthy eating habits.
Can this damage be reversed? The liver is amazing at reparing itself! The key is changing your habits: eating and exercise!
To read the full story:
http://www.sciencedaily.com/releases/2008/04/080430204519.htm#
Is what you're eating causing hepatotoxicity??

Unfortunately grapefruit is one of the substances that alters your liver enzymes. In fact it decreases their action. Can you imagine the effect that has on medications you take? If there are less enzymes to break down your medications, more of the drug sticks around. This increases your risk of toxicity and side effects. Grapefruit-drug interactions are known to be DEADLY!
The most well-known drugs that interact with grapefruit include: statins, heart rhythm medications, immunosuppressive drugs, and some blood pressure medications. However, many more medications such as over-the-counter remedies, anti-anxiety drugs, and antidepressants can be altered when grapefruit is consumed in conjuction with them.
When taking a medication, ask your healthcare provider about possible food and drug interactions. Various foods can have interactions with different medications.
Is this something specific to grapefruit? What about similar foods such as oranges and limes? Think about it!
Reference:
Blum, Alan. Mayo, Terri. Stump, Amy. http://www.aafp.org/afp/20060815/605.html. August 2006.
For more information on how grapefruit and other foods effect your medication visit:
http://www.lifesteps.com/gm/Atoz/ency/drug_metabolisminteractions.jsp
http://health.yahoo.com/nutrition-foodsafety/grapefruit-juice-and-medication/healthwise--tp23474spec.html
http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm096386.htm
Monday, October 19, 2009
Heart attack or Liver disease, Which would you prefer?
But at what price are these benefits realized? The American Cancer Society states that moderate consumption increases your risk of liver cancer in addition to many other forms of cancer. Dr. Jennifer Mieres of New York University School of Medicine noted the extent of liver damage associated with moderate alcohol consumption. With a damaged liver, imagine the possibilities of hepatotoxicity when you take any medication?
The key in protecting your heart is prevention. Lifestyle changes in diet and exercise have the same cardiovascular benefits as moderate alcohol intake. Unfortunately alcohol can be a convenient remedy to a complex problem, but it has its own health issues associated with it.
Think about your lifestyle and your medications, even if you just take Tylenol for headaches. Do the benefits outweigh the risks??
To read the full story visit: http://health.yahoo.com/news/healthday/drinkingyourwaytohealthperhapsnot.html
Sunday, October 18, 2009
What about alcohol?

Alcohol and illicit drugs can lead to three types of liver disease, fatty liver, alcoholic hepatitis, and alcoholic cirrhosis. Fatty liver is when fat cells accumulate in the liver. Most alcoholics experience this disease, and there are often no symptoms. However, some people do experience an uncomfortable feeling in their upper abdomen from the enlargement of the liver. After the cessation of drinking the liver will return to normal. Alcoholic hepatitis is when the liver becomes inflamed from the heavy drinking. Symptoms are numerous for this disease. Some of the symtoms include nausea, vomiting, loss of appetite, pain, fever, and jaundice. If mild, the disease could continue for years with gradual damage occurring, but in its most severe form, it can suddenly cause life threatening complications. Alcoholic cirrhosis is the most severe form of liver disease. Cirrhosis occurs when the normal liver cells are replaced with scar tissue cells. This disease is life threatening and is irreversible.
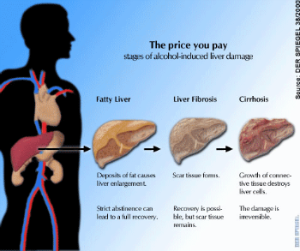
For most of us, small amounts of alcohol will not harm our liver. The standard for moderate drinking is one drink per day for women and two per day for men. However, for people that have existing liver conditions, take medication, or don't process toxins as well due to age related liver deterioration, even the smallest amounts of alcohol can have major effects on the health of their liver. Alcohol should never be taken with acetaminophen, or around the same time. Severe liver complications can arise from the mixture of these two toxins! It is CRITICAL to your safety to inform your health-care provider of ANY alcohol intake when taking medications or participating in procedures!
Think about your liver the next time you decide to have a drink!
Reference:
American Liver Foundation website. Last updated in 2007.
http://www.liverfoundation.org/education/info/alcohol/
How high of a price are you willing to pay to be thin?

Alli recently became available over-the-counter however, it is concerning that this potentially damaging product is readily obtained without health-care provider supervision. Most people believe over-the-counter products have few side effects in comparison to their prescribed counterparts. As we can see with this diet pill, that is often not the case.
Between 1999 and 2008, 32 episodes of serious liver injury associated with orlistat have been reported to the FDA. Symptoms associated with the use of these products include fever, jaundice (yellowing of the skin and eyes), right upper abdominal pain, nausea, loss of appetite, brown urine, muscle weakness, and fatigue.
Although orlistat has not been definately linked to hepatotoxicity, the FDA continues to investigate the effect of such therapies on the liver. When taking these medications, only take the prescribed amount, and be aware of hepatotoxic symptoms. If you do experience such effects, report these immediately to your health care provider as well as the FDA's MedWatch Adverse Event Reporting program.
Please visit the FDA website for more information and reporting procedures:
http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/DrugSafetyInformationforHeathcareProfessionals/ucm179166.htm
Are You at Risk for Over-the-Counter Hepatotoxicity?

1. Consumers perceive that OTC products are extremely safe and not likely to lead to serious toxicity. The marketing of OTC products emphasizes their safety and this perception may be reinforced by the availability of package sizes with large numbers of pills.
2. Consumers do not read the labels or follow the directions for use on OTC and prescription products.
3. Some of the prescription products are not adequately labeled to identify acetaminophen as an ingredient. Acetaminophen is often labeled as APAP on the prescription containers.
4. Consumers are not aware that acetaminophen can cause serious liver injury, in part because product labels do not adequately warn of this problem.
5. Consumers are not aware that acetaminophen is present in many OTC and prescription products and are not aware that they are exceeding the maximum daily dose.

6. Some populations (e.g., certain alcohol users and people with liver disease) are more susceptible to hepatic injury.
7. The symptoms of acetaminophen overdose may not appear for up to three days, so people may continue to take acetaminophen and increase the damage. The symptoms of liver injury may mimic the condition that they are treating (e.g., flu symptoms).
8. Because patients may not get adequate pain relief after taking the recommended dosage of acetaminophen, they may take more than the recommended amount or use other products that also contain acetaminophen.
9. Patients develop tolerance to narcotics and need to increase the dose of prescription combination products. If they do this on their own, they may not realize that they are increasing the dose of acetaminophen to toxic levels.
10. Combination narcotic products are commonly used because of limited non-narcotic options and greater restrictions on availability for higher scheduled single-ingredient narcotic analgesics.
For more information, please read the full report by The Acetominophen Hepatotoxicity Group.
References:
The Acetaminophen Hepatotoxicity Working Group Center for Drug and Evaluation Research, F. D. (2008). Recommendations for FDA Interventions to Decrease the Occurence of Acetaminophen Hepatotoxicity.
FDA and Acetaminophen…… Is it ENOUGH???
 It is important to note the that initiation of a strategy to eliminate hepatotoxicity can decrease the number of unintentional overdose cases. There is no one factor that is linked to hepatotoxicity with acetominophen; many conditions often contribute to this problem.
It is important to note the that initiation of a strategy to eliminate hepatotoxicity can decrease the number of unintentional overdose cases. There is no one factor that is linked to hepatotoxicity with acetominophen; many conditions often contribute to this problem. Below is a list of ways that the FDA has recommended to reduce the occurrences of acetominophen hepatotxicity:
1. Enhance public education efforts (also may reduce intentional overdoses)
- Develop concise, clear messages
- Increase partnerships with other governmental agencies, health professionals, industry, consumers, and media
- Prominently display the name acetaminophen on principal display panel
- Include warning that taking more than recommended amount may cause severe liver injury and should not be used with other products containing acetaminophen (on the container and box)
- Include warning about need for prompt medical attention after acetaminophen overdose even when no symptoms of a health problem are present
- Include warnings for people with liver disease and alcohol users
4. Limit the tablet strength for immediate-release forms to 325 mg, single adult dose to 650 mg, and extended-release forms commensurate with total daily dose.
5. Limit options in pediatric liquid formulations
- Limit pediatric liquid formulation to one mid-strength concentration
- Require that a measuring device be included in each package
- Include dosing instructions for children under 2 years if accurate dosing instructions can be determined.
The Acetaminophen Hepatotoxicity Working Group Center for Drug and Evaluation Research, F. D. (2008). Recommendations for FDA Interventions to Decrease the Occurance of Acetaminophen Hepatotoxicity.
Friday, October 16, 2009
Seeing through the confusion, should we stop taking Tylenol/acetaminophen?
It could take quite a while for the FDA to definatively decide about new dosages of acetaminophen. In the mean time, people and doctors are baffled. The vote that took place in June from the panel was for the reduction of the current recommended dose of acetaminophen. Another vote included making certain products containing 500 mg or more available through prescription only. Once a decision from the FDA is reached, it could still take years for it to actually take effect.
Acetaminophen related hepatotoxicity is a major concern for the FDA because it is one of the most commonly used pain relievers on the market. In 2004, they released a campaign to inform the public about the dangers of acetaminophen misuse. Some believe that it has not been fully effective in reducing the amount of toxicity because it is still at the top of the list of drugs that commonly cause liver damage.
Some have countered that doctors and pharmacists need to be the ones to take note of the dangers related to acetaminophen. Currently, the recommended daily dose is 4 g, but pharmacists say that they have seen doctors prescribe much more than that. Who's responsibility is it to reach the consumer?
In the end, what has one representative said about the safety of the current dose? People have no need to worry, that as long as it is used correctly 4 grams per day should be just fine.
Is acetaminophen (brand name, Tylenol) cause for concern? Next time you reach into the medicine cabinet, what will cross your mind?
For more details, view the article:
http://libproxy.uta.edu:2066/login.aspx?direct=true&db=rzh&AN=2010377171&site=ehost-live
This is a video with the discussion of safety:
http://www.youtube.com/watch?v=K7WCSwEWjdk
Reference:
Traynor, A., (2009). FDA’s acetaminophen meeting sparks confusion.
American Journal of Health-System Pharmacy, 66(16), 1422, 1425-6.
Retrieved from: http://libproxy.uta.edu:2066/login.aspx?
direct=true&db=rzh&AN=2010377171&site=ehost-live
Tuesday, October 13, 2009
Is Tylenol a threat?
When using these medications, it is important to check the labels for ingredients. Certain medications, including cold and flu remedies, contain acetaminophen. Many people take multiple drugs at one time that each contain acetaminophen. This results in an acetominophen overdose that causes dangerous hepatotoxicity and can be fatal! Some medications list acetaminophen as APAP, it's chemical name abbreviated. Recognizing this is critical in keeping your liver safe from acetominophen hepatotoxicity!!
In 2007, reports from the American Association of Poison Control Centers stated that:
-18,108 people in the US had been poisoned by taking acetaminophen products like Tylenol
-75,814 people in the US had been poisoned by medication containing acetaminophen
-1,239 reports from poison resulted in death
-348 of these deaths were a result from acetaminophen
These are alarming statistics to see, especially since these drugs are so easily available. Due to the liver damage caused by acetaminophen, it has been suggested to the FDA that the recommended dosage be reduced. New recommendations are going to be voted on in the future, and they will probably be much lower than the current standard of two tablets in a dose.
To view the article follow this link:
http://www.cleveland.com/healthfit/index.ssf/2009/09/is_two_too_many_fda_is_reconsi.html
Reference:
Suchetka, D., (2009). Is two too many? FDA is reconsidering the proper dosage levels for OTC painkillers. Northeast OH Healthy Living and Medical Consumer News. http://www.cleveland.com/healthfit/index.ssf/2009/09/is_two_too_many_fda_is_reconsi.html
Monday, October 12, 2009
Who is Helping to Prevent Hepatotoxicity?
Government agencies play a pivotal role in the prevention and regulation of drugs linked with liver injury. FDA intervention strategies can significantly decrease the number of cases of drug overdose leading to liver injury from over-the-counter and prescription drug products. Organizations such as the Center for Drug Evaluation and Research (CDER), Office of Surveillance and Epidemiology (OSE), American Association for the Study of Liver Diseases (AASLD), Drug-Induced Liver Injury Network (DILIN), National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), and Pharmaceutical Research and Manufactures of America all recognize the severity of hepatoxicity.
Can we do more to reduce drug-induced hepatotoxicity? How can we make people taking these medications active in this process?
References:
John R. Senior, M. L. (2009, July 31). Drug-Induced Liver Toxicity. Retrieved October 11, 2009, from Food Drug Administration: http://www.fda.gov/Drugs/scienceresearch/researchareas/ucm071471.htm
Melissa Palmer, M. (n.d.). MEDICATIONS AND THE LIVER/HEPATITIS. Retrieved October 17, 2009, from http://www.liverdisease.com/medications_hepatitis.html
Hepatotoxicity: Where We've Been, Where We Are, Where We're Going
HISTORY
Hepatotoxicity dates back to nearly 100 years. Just recently, they have discovered that some of the most common drugs can cause liver injury. These drugs are currently well known to the public and used by a majority of the population. The drugs in question are known as non-steroid anti-inflammatory drugs (NSAIDS), statins, anti-diabetics, and acetaminophen.
- Acetaminophen was known to cause liver damage in 1943, if not taken in therapeutic range. Even when taken appropriately, can cause hepatotoxicity if taken with other medications.
- The anti-diabetic drug, troglitazone, was the first drug to bring light to hepatotoxicity and the long-term causes it can have on the liver.
TODAY
Hundreds of articles exist on the effects of drugs on the liver. The drug industry has been hit hard with removal of drugs from the market that cause liver damage. Liver hepatotoxicity is one of the most common reasons that drugs are withdrawn from the public's use. Even doses in the therapeutic range, have been known to cause toxicity of the liver. Although these drugs were known to work effectively, they were shown to cause severe liver damage, liver transplant, or death.
In 2006, the FDA added guidelines to follow if the immune system falls below a certain range. Liver enzymes should be tested in order to keep patients safe. This is currently the only way to diagnosis liver hepatoxicity, at least the most efficient way.
Recently, the public has become aware of acetaminophen related hepatotoxicity. One controversy on OTC drugs is the drug-drug interactions. The public is not as educated as they should be on the ease of overdosing on OTC drugs. As healthcare professionals, it should be an important goal to educate patients on liver safety.
WHAT THE FUTURE HOLDS
Drug companies will have a never-ending battle with regulating the side effects of drugs. It could be considered a catch 22, drugs can dramatically improve ones life and at the same time be a silent killer. The FDA has taken steps to educate the public on these drugs, but the warnings do not seem to be eye catching enough.
Studies show that there is a possible genetic link to drug induced liver injury. Science Daily (2009) studies show that acetaminophen is toxic if taken in high doses, but a link exists showing that toxicity is more likely due to genetics. The study suggests that finding the genetic marker will help create safer drugs in the future. Studies also suggest that some patients become more susceptible due to their environmental factors.
Current preclinical and clinical liver safety testing does an excellent job in keeping the public safe from drugs capable of causing various forms of liver injury.
In any case, the FDA will constantly be on alert for drugs and their contraindications for hepatotoxicity. Healthcare professionals will need to be aware of the ever-changing revisions to drugs and safety changes.
References:
Lucena, M., García-Cortés, M., Cueto, R., Lopez-Duran, J., & Andrade, R. (2008). Assessment of drug-induced liver injury in clinical practice. Fundamental & Clinical Pharmacology, 22(2), 141-158. http://search.ebscohost.com/
Watkins, P. (2005). Idiosyncratic liver injury: challenges and approaches. Toxicologic Pathology, 33(1), 1-5. http://search.ebscohost.com/
Fontana R, Watkins P, Bonkovsky H, et al. Drug-Induced Liver Injury Network (DILIN) prospective study: rationale, design and conduct. Drug Safety: An International Journal Of Medical Toxicology And Drug Experience [serial online]. 2009;32(1):55-68. Available from: MEDLINE
Ipswich, MA. Accessed October 16, 2009.Lee W, Senior J. Recognizing drug-induced liver injury: current problems, possible solutions. Toxicologic Pathology [serial online]. 2005;33(1):155-164. Available from: MEDLINE, Ipswich, MA. Accessed October 16, 2009.009.





